In recent years, many new devices have attempted to solve the challenges associated with Neuroendovascular aneurysm treatment technology more specific, wide-neck aneurysm neuroembolization. This has led to a plethora of neuroendovascular interventional options based on aneurysm location, size, parent vessel configuration, hemodynamic patterns, etc. One such novel device, the Comaneci™ embolization-assist device from Rapid Medical, aiming to make the such challenging intracranial aneurysm treatment decision tree easier, as it enables coil-assisted embolization ruptured or unruptured, sidewall or bifurcation wide-neck aneurysms, does not block blood flow in the parent vessel, and without the need for long-term dual antiplatelet therapy.
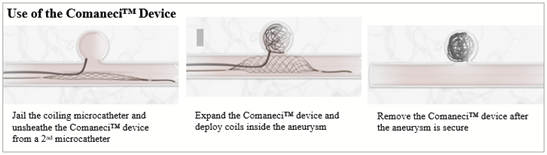
The Comaneci embolization-assist device features an expandable radiopaque remodeling mesh that conforms to the vessel wall, providing temporary coverage of the aneurysm neck with constant perfusion. With a thumb-controlled slider (fig.1), Comaneci expands in the parent vessel across the aneurysm neck, providing a buttress for coil embolization (fig.2). The device is then collapsed and removed at the conclusion of the procedure, allowing aneurysm embolization in without permanent stent placement and long-term dual antiplatelet therapy.

Mohammad El-Ghanem
College of Medicine, Tucson Arizona
Figure 1
Figure 2
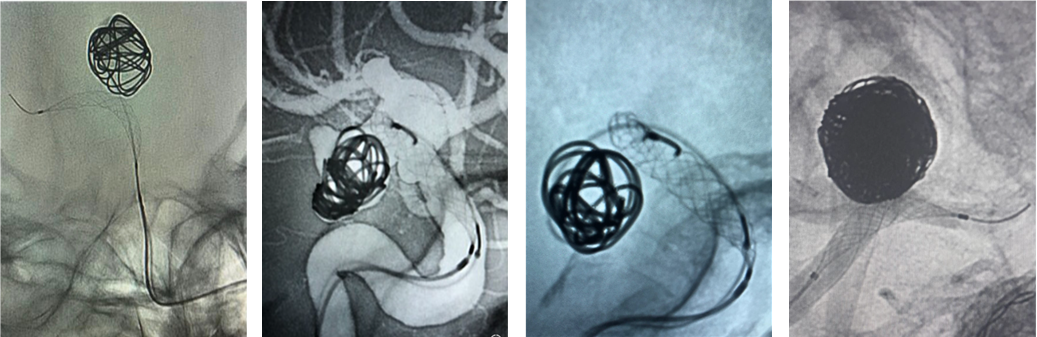
Though recently FDA cleared, the Comaneci device has been used in over 4,000 wide-neck coil embolization procedures worldwide (Fig3.).
Though not comparative, evolving data1,2 shows that the Comaneci-assisted coiling is a feasible and safe way to treat wide-neck aneurysms. The major clinical advantage of this device is that it could be applied in ruptured aneurysm cases without the need of antiplatelet therapy.
The Comaneci device is available in 3 sizes; Comaneci and Comaneci Petit are delivered through an 0.021-inch microcatheter and the Comaneci 17 is delivered via an 0.017-inch microcatheter.
Figure 3